 Lithium has about half the density of water, giving solid sticks of lithium metal the odd heft of a light-to-medium wood like pine. The fresh metal has a silvery-white color which only remains untarnished in dry air. Lithium is soft enough to be cut with a knife, though this is more difficult than cutting sodium. These and many other properties attributable to alkali metals' weakly-held valence electron are most distinguished in lithium, as it possesses the smallest atomic radius and thus the highest electronegativity of the alkali group. As a result, lithium is easily deformed, highly reactive, and has lower melting and boiling points than most metals. Like other alkali metals, lithium has a single valence electron which it will readily lose to form a cation, indicated by the element's low electronegativity. So the lithium is the least reactive and Cesium is the most reactive. The lower the ionization energy the more reactive these metals. They are very soft metals that can be cut with a knife. They have very low ionization energy and give up their outer s 1 electron very easily. They must be stored under oil or they will quickly oxidize. Group 1 Metals are the most reactive metals on the periodic table and do not exist free in nature. It is important to recognize which numbering system is being used and to be able to find the number of valence electrons in the main block elements regardless of which numbering system is being used.Group 1 Group 2 Group 3-12 Group 15 Group 16 Group 17 Group 18 You will come across periodic tables with both numbering systems. In this numbering system, group 1A is group 1 group 2A is group 2 the halogens (7A) are group 17 and the noble gases (8A) are group 18. The elements in this group are also gases at room temperature.Īn alternate numbering system numbers all of the \(s\), \(p\), and \(d\) block elements from 1-18. We will learn the reason for this later, when we discuss how compounds form. These elements also have similar properties to each other, the most significant property being that they are extremely unreactive, rarely forming compounds. This group contains very reactive nonmetal elements. Group 7A (or 17) elements are also called halogens. It is important to recognize a couple of other important groups on the periodic table by their group name. Remember, Mendeleev arranged the table so that elements with the most similar properties were in the same group on the periodic table. The same pattern is true of other groups on the periodic table. Once again, because of their similarities in electron configurations, these elements have similar properties to each other. Group 2A is also called the alkaline earth metals. Although most metals tend to be very hard, these metals are actually soft and can be easily cut. 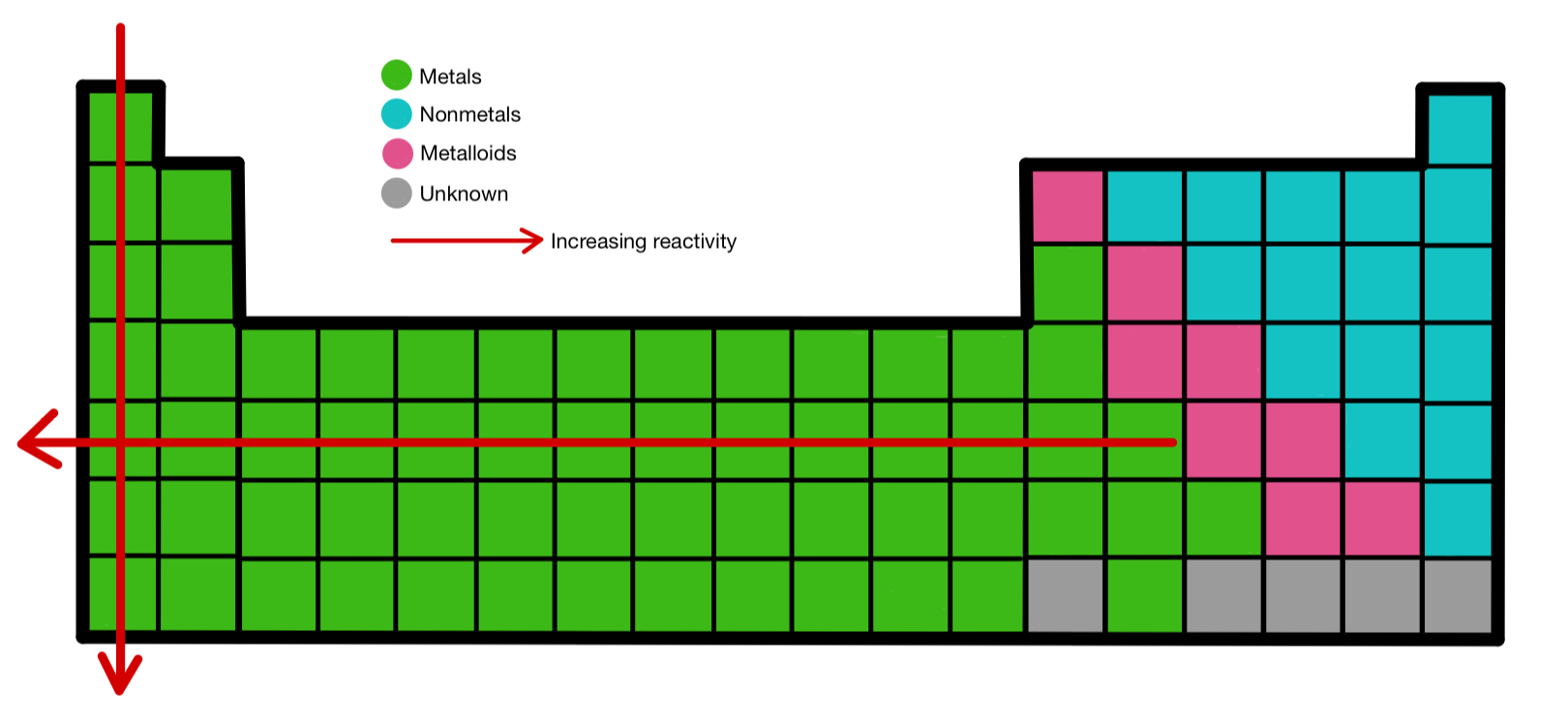
Group 1A is also known as the alkali metals. 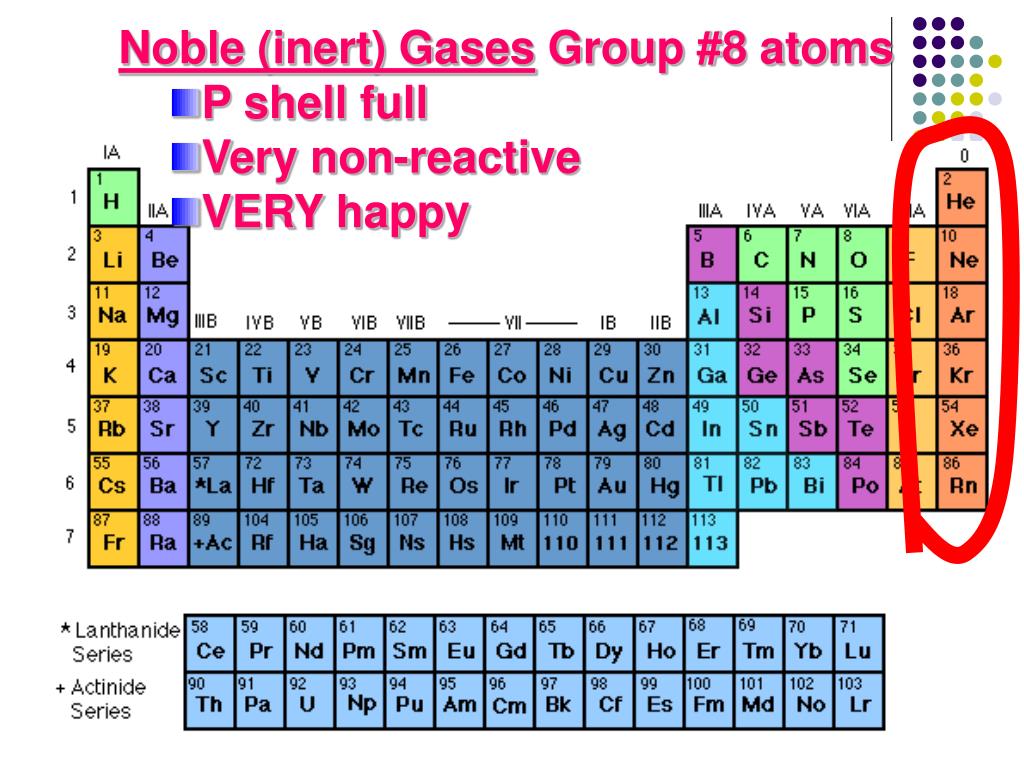
Because of their similarities in their chemical properties, Mendeleev put these elements into the same group. The elements in 1A are all very reactive and form compounds in the same ratios with similar properties with other elements. This is what causes these elements to react in the same ways as the other members of the family. 
All of the 1A elements have one valence electron. A group is a vertical column of the periodic table. Remember that Mendeleev arranged the periodic table so that elements with the most similar properties were placed in the same group. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
